In this tutorial we will calculate the density of water using a balance and a metered water container.
- Weight the mass of the plastic cylinder.

- Pour 60ml water into the cylinder and weight once again the mass. The difference between the final and the initial mass is the mass of water.

- The density formula is:
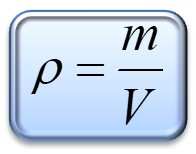

By using the values you weighted you can find that:
- Weighted mass of water= 107–47= 60g
- Water volume= 60 ml = 60 cm³
- So, ρ = 60/60 = 1 g/cm³
- Repeat the procedure by using different kinds of liquids and compare the different densities.
Question 1
Which of the following properties is fundamental?
- force.
- mass.
- acceleration of gravity.
Question 2
Which of the following properties is derivative?
- mass.
- length.
- weight.
Question 3
Which of the following units is fundamental?
- kilogram Kg
- gram g
- Newton N
Question 4
The mass is affected by:
- The shape of the object
- The color of the object
- The volume of the object
- None of the above
Question 5
If the density of water is 1g/cm3 and the density of ice is 0,92g/cm3, which has more volume, 1kg of water or 1kg of ice?
- Ice.
- Water.
- They have the same volume.
Question 6
Two solid objects A and B have densities ρΑ>ρΒ and the same mass. So:
- VA > VB
- VA < VB
- VA = VB
Question 7
A piece of cork is cut into two equal pieces. The density of each piece is:
- the same with the initial piece.
- twice the density of the initial piece.
- the half of the density of the initial piece.
End of tutorial