In this experiment we examine the states of mater using Polytech’s BT11 Teacher’s STEM Lab Kit.
Method
We use water in solid and liquid forms and by applying heat we observe the change in the states of matter from solid to liquid and later to gas. During the whole process we monitor the temperature change of the matter over time on a real time graph.
Contents
- Introduction
- Phases – Material characteristics
- Matter change of state through Energy
- Setup Instructions
- Connections
- Usage Instructions
- Questions
Resources
Materials
- Tap Water
- Ice cubes
Hardware & Software
- Windows PC with Bluetooth
- ilab for Windows Application
Safety Notes
Take all necessary precautions when using open flame equipment. Make sure that heat does not affect or damage the equipment used.
1. Introduction
Phases – Material characteristics
Matter comes in 4 different structures.
- Solid objects have definite form and shape.

- Liquids have definite form and the take the shape of the container they are in.

- Gases have no definite shape nor form and they take the form of the container they are in.

- Plasma is a superheated gas where atoms lose electrons, creating a mix of charged particles, found in stars and lightning.

In all of the forms and shapes matter can be defined or measured by some basic characteristics as follows:
- Volume
- Mass
- Density
- Temperature
- Specific gravity
- Specific volume
Matter change of state through Energy
Matter changes states through heat transfer. Adding heat increases particle energy, causing melting or vaporization. Removing heat decreases energy, leading to freezing or condensation. Heat gain or loss drives these transitions.
This experiment will demonstrate the change of state of water from solid to liquid and finally gas by applying heat.
2. Setup Instructions
- Assemble the retort stand with two metal rods connected with a boss perpendicular to each other.

- Adjust the clamp on the horizontal rod

- Fix the temperature sensor probe using the retort clamp facing downwards and rotate the stand so that the sensor probe is in distance from the BT11 base.

- Place the ice cubes in the beaker. Add some tap water.
- Place the beaker on the heating plate
- Place the heating plate under the temperature sensor probe. Make sure the probe is inserted thoroughly in the ice and water mix, in a safe distance from the bottom of the beaker (not touching).
Hint: Adjust the height of the horizontal rod carying the temperature sensor pdobe to find the ideal position.

3. Connections
- Connect the temperature sensor to the BT11 with UTP cable.

- Connect the BT11 to the computer with wireless connection. (For further instructions consult the “Operating BT11” manual).
- Turn on the BT11 datalogger by pressing the button on the right side.

4. Usage Instructions
- Run the iLab application and select Start
 .
.
- On the screen that appears press the
 icon
icon
- On the window Select display mode that is now open, select the following:
- Turn on the alcohol lamp.
- Observe the graph as the temperatures are recorded. When the water starts boiling, continue recording for another 5 minutes. Press “Stop (F8)”
 to stop recording. The graph will look as follows:
to stop recording. The graph will look as follows:
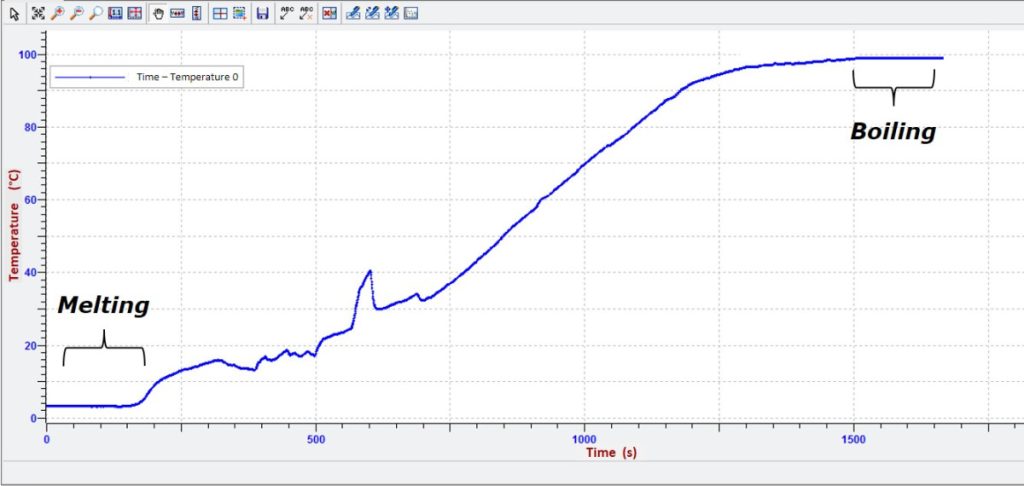
The temperature of the ice (solid) increases until it reaches 4 °C. At this point, we observe the phenomenon of melting (during which the solid and liquid water coexist) and the temperature remains constant until the entire ice is converted into liquid.
Then the temperature of the water increases and part of the liquid is converted into gas (evaporated). When the water reaches near 100 °C, we observe the phenomenon of boiling with bubbles rising to the surface (boiling, evaporation). Throughout the boiling, the temperature remains constant until the whole liquid is converted into gas.
Note: The melting-freezing temperature and the boiling temperature of the water do not result exactly equal to the values 0 °C and 100 °C, respectively, in all the experiments, but they depend on the purity of the water, the ambient pressure, the altitude at which the experiment is conducted and other factors.
Question 1
Which of the following phase changes is right?
- Liquefaction solid -> liquid
- Melting gas -> liquid
- Freezing liquid -> solid
- Volatilization solid -> gas
Question 2
Rain, snow and hail result from the _____ of water.
- transpiration
- precipitation
- runoff
- condensation
Question 3
According to the model, where do you observe evaporation?
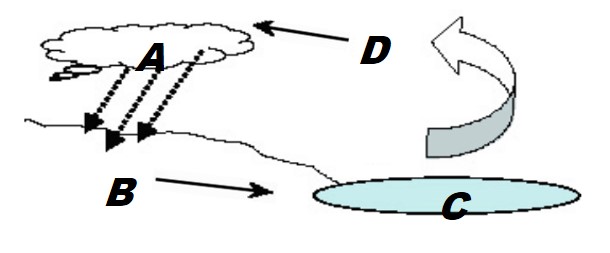
- from Α to Β
- from Β to C
- from C to D
- from D to Α
Question 4
According to the model, where do you observe condensation?

- from Α to Β
- from Β to C
- from C to D
- from D to Α
Question 5
According to the model, where do you observe precipitation?

- from Α to Β
- from Β to C
- from C to D
- from D to Α
End of tutorial
