In this tutorial we will explore the main theory on various measurements such as volume, mass, density, temperature, pressure, specific weight, specific gravity and technology of measurements.
Course contents
In this tutorial we will study:

1. Introduction
Phases – Material characteristics
Matter comes in 3 different structures.
Solid objects have definite form and shape.

Liquids have definite form and the take the shape of the container they are in.

Gases have no definite shape nor form and they take the form of the container they are in.

In all of the forms and shapes matter can be defined or measured by some basic characteristics as follows:
- Volume
- Mass
- Density
- Temperature
- Specific gravity and Specific volume
2. Volume
Volume is the quantity of three-dimensional space enclosed by a closed surface, for example, the space that a substance (solid, liquid, gas, or plasma) or shape occupies or contains.
So, the formulas we learned in geometry for calculating volume are the basis to start describing the shape and the form of the matter.
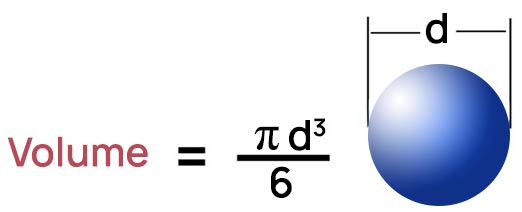
If a liquid is in a container, its volume is the part of the container that the liquid occupies.

If a gas is in a container we know it occupies the whole space of the container so the volume of the gas is the whole container.

3. Mass
The subject of mass was covered in Mechanics but let us review our knowledge on the subject.
Mass is not the weight of the object.
The balance shows 96.7gr, that measures the mass of it. The weight of it is the mass multiplied by the gravitational acceleration and it is measured in Newton.
Mass is the amount of matter that a body contains and also a measure of its resistance to change of motion (Inertia).
Mass is different from weight, which is a measure of the attraction of the earth for a given mass.
The standard unit of mass is kg.
The standard unit of weight is N.
4. Density
Density is a measurement of how heavy something is for its size.
Mathematically, density is the ratio of mass divided by volume.
For example, a baseball and a steel ball having the same diameter d may occupy the same amount of space (that is have the same volume), but the steel ball has more mass, therefore it is more dense than the baseball.
Density is an important concept in science, and it has practical applications in all structural engineering fields and material sciences.
The mass density or density of a material is defined as its mass per unit. The symbol used for density is the Greek letter ρ.
Density ρ is defined by the equation:
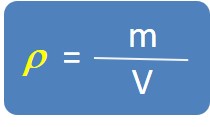
The unit of density is kg/m³ or g/cm3.
From geometry, the metal cylinder has volume V= π x d2 x h/4
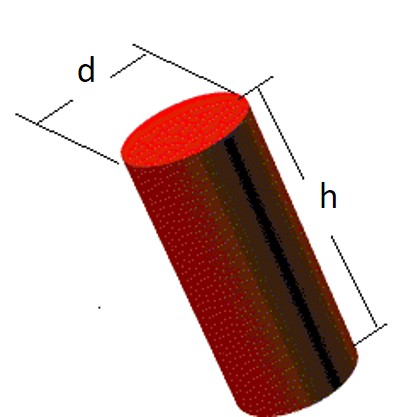
Lets assume the volume of this cylinder is 40cm3 and the balance shows that its mass is 108 gr. That gives us:
Density ρ=108g/40cm3=2.7g/cm3=2,700 Kg/m3
Density allows us to identify the metal. Aluminum has this value of 2,700 Kg/m3 of density as we will see later.

Question 1
What is density?
- The volume of a substance
- The weight per unit of volume of a substance.
- The mass per unit volume of a substance.
- The volume per unit of mass of the substance.
Density identifies substances and differentiates objects. Two objects of equal volume having different masses they have different density. The same happens when 2 objects have equal mass but different volume.
Look at the photo of the 4 metal rods. They all have equal mass. Note the difference in volume. They are different metals, they have different internal molecular structure and they have different densities.

Just to make it more obvious these cylinders have also the same diameter (d), so the height h difference depicts accurately the relationship of their densities since diameter (d) and mass (m) are all equal the only variant is the height h.
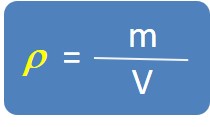
Question 2
Which is heavier, a kilogram of iron or a kilogram of aluminum?
- One kilogram of iron
- One kilogram of aluminum
- They both have the same mass so they weight the same
- None of the above
4.1. Measuring Density
So far we have practicing with solid matter and defined shapes and forms.
Before we explore how we can measure density in irregular solid shapes let us examine density of liquids and take again as our study subject the most known liquid matter, the WATER.
The density of water is 1gr/cm3
As we said before solids have higher density than liquids, so we would expect the same for water. This is not the case though. The solid water or Ice has less density than liquid water. That is why the also Ice floats on water.

Ice
The density of ice is 0.9167 g/cm³ at 0 °C, whereas water has a density of 0.9998 g/cm³ at the same temperature. Liquid water has 1.00 g/cm³, at 4 °C and becomes less dense as its the freezing point is reached.
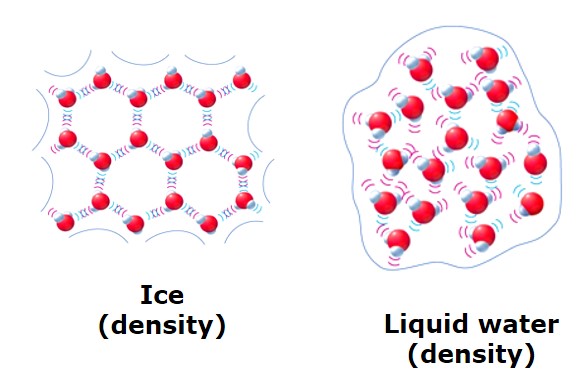
The fact that water expands upon freezing causes icebergs to float. The expansion during the phase change from liquid to solid form is in contrast to most of the other substances.
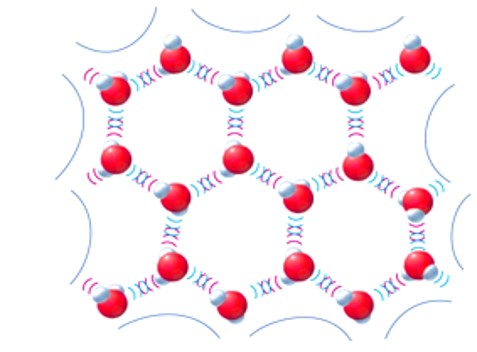
The expansion of the volume in ice comes from the fact that water crystallizes into an open hexagonal form. This hexagonal form of the ice takes more space than the liquid water. Look at the molecule of water and ice. See the empty spaces forming when ice is form in its 6 angle (hexagonal) shape.
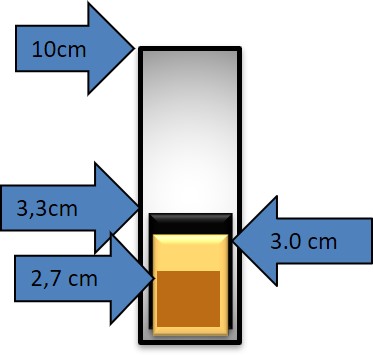
4.2. Measuring the density of a liquid
To calculate the density of a liquid, we have to measure the mass of the container that we will use, a graduated tube.
Next, we fill the container with the liquid up to a certain point of known volume for example 150ml or V=150 cm3
We measure the mass of the container with the liquid.
The Volume of the liquid is known, and the mass of the liquid can be calculated from the difference between the two mass readings.
m = 255g – 75g = 180g
Now, knowing the mass and the volume we can calculate the density of the liquid.
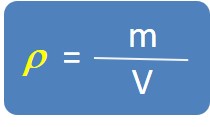
180g/150cm3=1.2g/cm3
4.3. Density of irregular shape solids
If a solid object of volume V is irregular (not defined geometric shape) we can measure its density using a graduated tube or flask or use the Archimedes principle, which will explore in matter 1 topic later too
We can immerse the object in the graduated tube of known volume of water. Its volume will be equal to the increase of the level of water. Then we measure its mass and we can calculate its density.
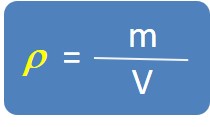
To measure the volume V of an irregular object we can also use Archimedes overflow container and a displacement beaker. The volume of the water over-flown into the displacement beaker is equal to the volume V of the object.
Density of gases also is calculated by the same formula.
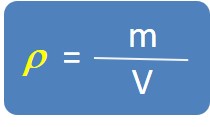
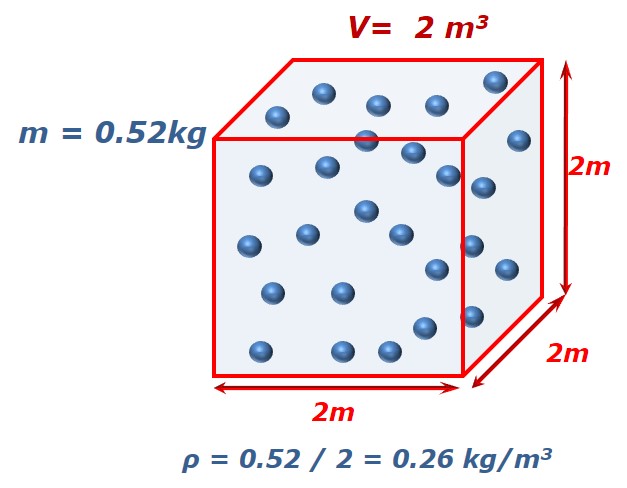
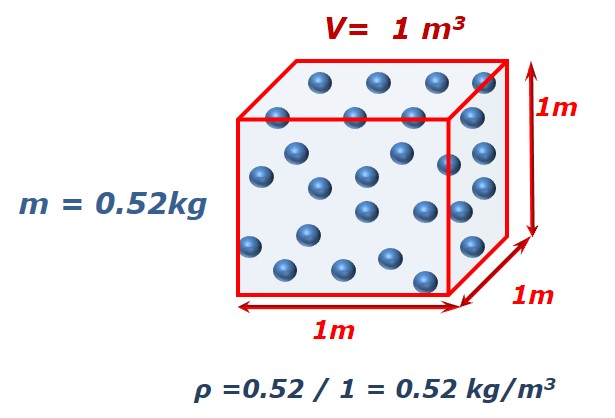
Since density is defined to be the mass divided by the volume, density depends directly on the size of the container in which a fixed mass of gas is confined. Lets us take this example:
We have 26 molecules of a X gas. Each molecule has a mass of 20 grams (0.02 kilograms), so the mass of this gas is 0.52 kg. The volume of its container lets say is 2m3. If we can reduce the size of the container to 1 m3 then density will be doubled.
Question 4
What is the SI unit used to measure density?
- kg/cm³
- g/m³
- m/ml
- kg/m³
5. Temperature
Temperature is a measure of the kinetic energy of the molecules of a substance
The kinetic theory states that matter consists of small particles, molecules and atoms. These particles possess kinetic energy which causes them to vibrate and move in various speeds and directions.
Temperature is measured using Thermometers which use some types of fluid, generally mercury. Thermometers measure temperature in various scales. The most common are Kelvin, Celsius, and Fahrenheit.

Thermometers rely on the simple principle that a liquid changes its volume relative to its temperature. Liquids take up less space when they are cold and more space when they are warm (this same principal applies for gases)
By heating the liquid in the thermometer, it expands and moves up in the tube.
5.1. Density and temperature
Density and Volume are reverse proportional.
Temperature changes the volume of any matter.
That means temperature changes the density of matter when is increased or decreased.
The more kinetic energy a substance has, the warmer it will be and the faster particles will be moving, which reduces the density of the substance.
When temperatures increase, objects expand and become larger and therefore the density decreases.
When temperatures decrease, objects condense and become smaller so density increases. There are always exceptions as we saw in liquid water and ice.
It is therefore very important to know the temperature at which a density has been measured. Recorded densities for different substances always state the temperature in which density was measured.
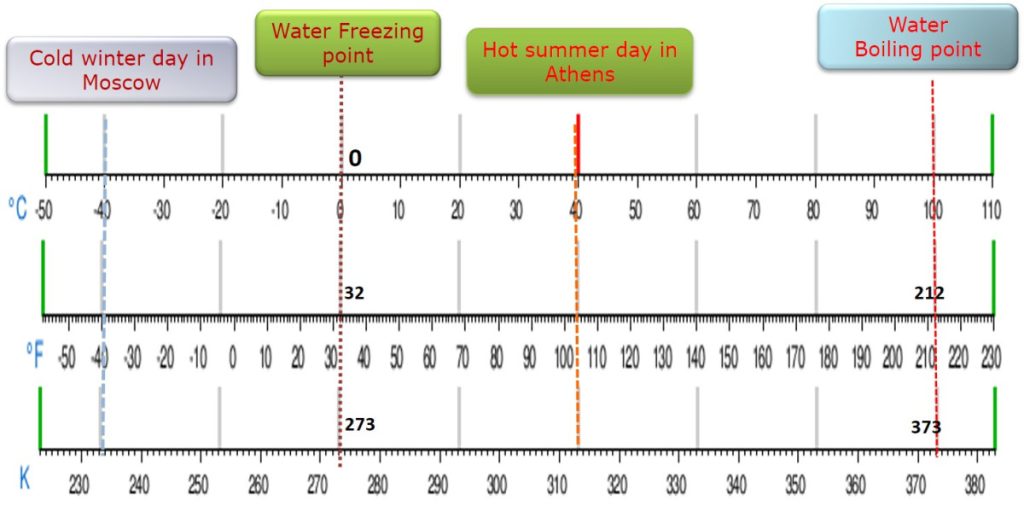
5.2. Temperature scales
Thermometer scales are different. Let us take a look at them and see their relationships at some temperature measurements in Celsius (°C), Fahrenheit (°F) and Kelvin (°K) scales.
6. Specific Weight
Specific weight (also known as the unit of weight) is the weight per unit of volume of a substance. The symbol of specific weight is γ (the Greek letter Gamma).
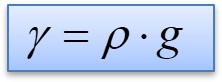
Where:
- γ the specific weight of the material, in N/m3
- ρ the density of the material in kg/m3
- g the gravitational acceleration in m/s2
The relationship between Specific weight and density is the same between mass and weight. We should not confuse them also.
In fluids, specific weight represents the force exerted by gravity on a unit volume of a fluid.
Specific weight is calculated measurement
The SI for specific gravity is N/m3.
The table depicts the Specific weight of water at sea-level in different temperatures. Do you observe that water at 5°C has more specific weight than in 0°C ?

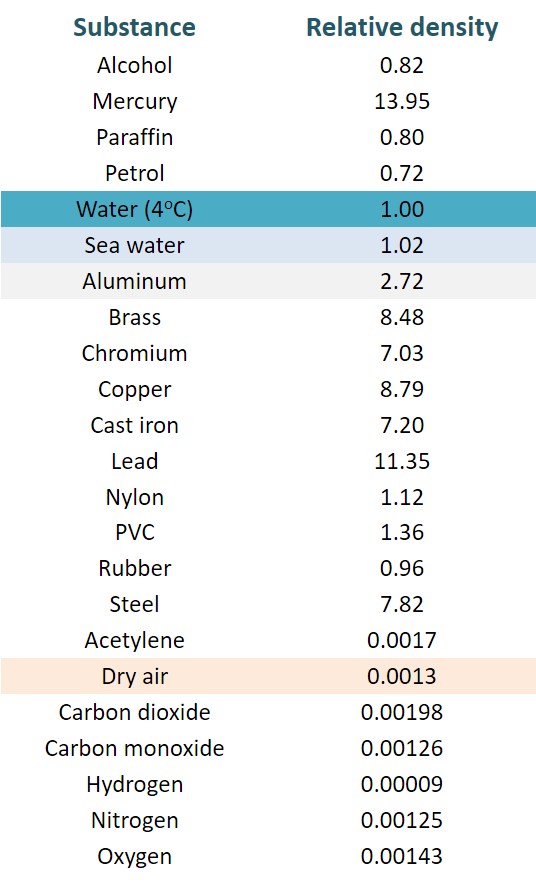
7. Specific Gravity
Specific gravity or also called Relative density of a substance is the ratio of the of the substance density to the density of water at 4oC.
Specific gravity, as it is the ratio of densities, has no dimension. It just an index number and has no SI unit of measurement. The specific density of Ice is less than 1, that is why it floats on water.
Specific Gravity can be expressed as SG = ρ/ρH2O, where:
- SG the specific gravity
- ρ the density of fluid or substance (kg/m3)
- ρH2O the density of water (kg/m3) at 4°C
Question 5
“If the specific gravity of a substance is greater than 1, this substance will float on water at temperature of 4°C”. Is this statement True or False?
- True
- False
8. Technology of measurements
During this tutorial, we presented what we can call classical ways of measuring the characteristics of Matter, as for example the Thermometer.

Temperature or pressure are basic parameters in technology. The modern digital revolution is offering us complicated systems which integrate multiple measurements, in multiple points in order to be controlled, ranging from our simple electrical appliances and our cars to huge industrial systems.

Having calculated the volume and knowing the possible error of each measurement of the sides, how we calculate the error rate in the calculation of the volume?
These control systems, called Automatic control systems, use data logging systems to measure very detailed data and respond immediately to any adjustments needed on a process.
When you adjust the temperature in you electric furnace, your refrigerator, your iron or when you are looking at you car oil pressure and temperature, you are simply controlling or monitoring the temperature or pressure in a process.

A basic training data monitoring system is usually comprised of a Data logger (collector of data) to store on line our measurements.

Different types of sensors connected to the data logger, depending on the types of measurement we need i.e. Temperature measurements and pressure measurement.

…and computer system where we analyze the data collected, make graphical representations equate to formulas etc..
End of tutorial